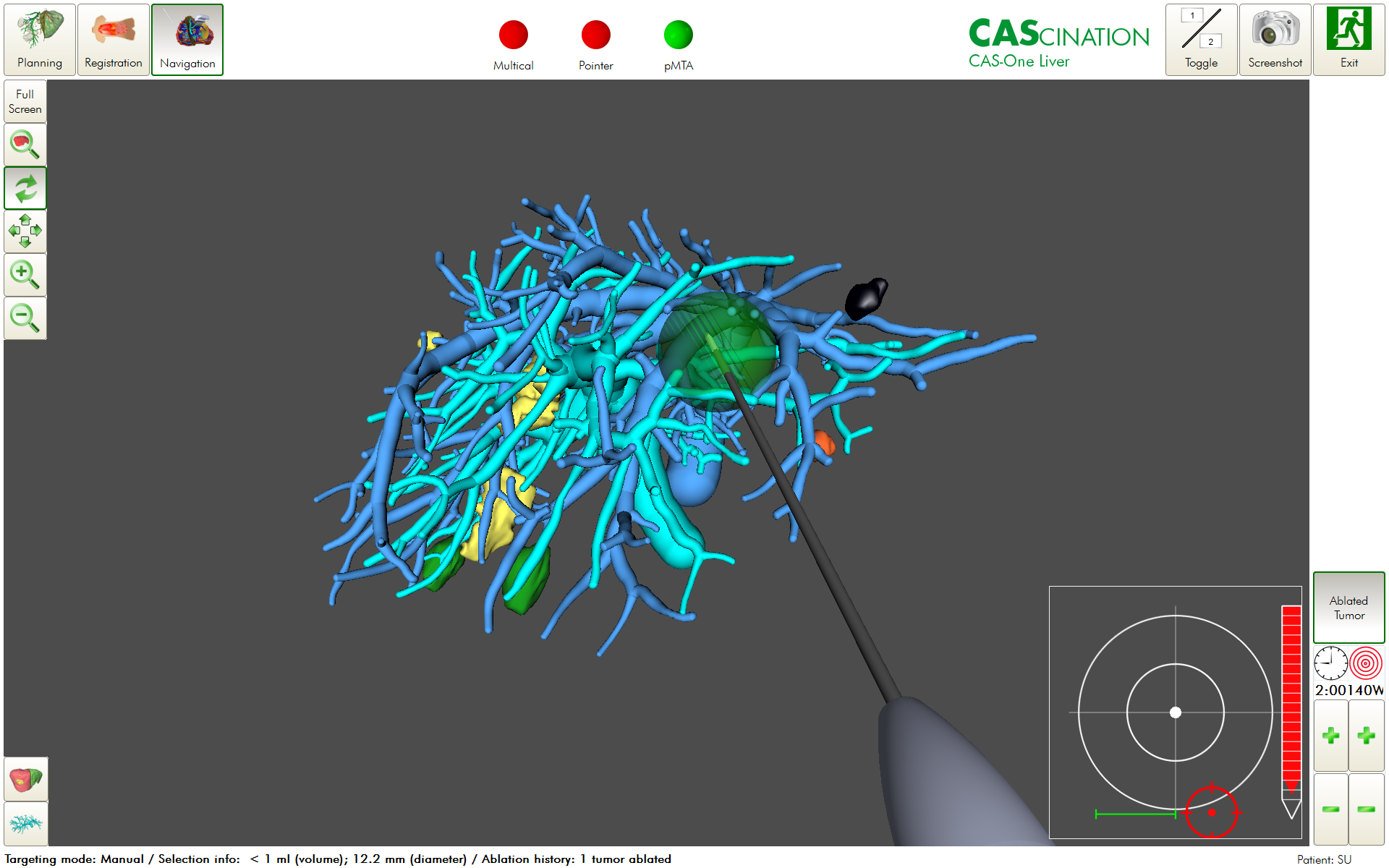
Despite major advances in modern medicine, pancreatic cancer remains for the most part a death sentence. If we are to change the trajectory of pancreatic cancer, early diagnosis is probably the most effective tool at hand. Patients diagnosed with pancreatic cancer at an early stage have the best chance of curative treatment and long-term survival. For example, localized cancer confined to the pancreas has a 5-year survival rate of 42%1. Once the disease has spread to regional structures or lymph nodes, the 5-year survival drops to 14% and only 3% of those diagnosed with distant metastases survive beyond 5 years. However, developing early detection methods for pancreatic cancer remains an elusive task and still today, only around 13% of patients have their tumour detected at a localized stage. The striking difference in survival between early- and late stage tumours has spawned decades-long efforts to find biomarkers that will enable earlier detection of pancreatic cancer.
Figure 1. Memorial stone for American actor Patrick Swayze
The US National Institutes of Health (NIH) defines a biomarker as “a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention.”2 Carbohydrate antigen 19-9 (CA 19-9), discovered in 19793, is the only FDA-approved serum biomarker for pancreatic cancer. CA 19-9 has a sensitivity of 79-81% and specificity of 82-90%4. However, CA 19-9 has a limited sensitivity in early-stage disease. False positive values may occur in patients with benign biliary obstruction and several inflammatory conditions in the pancreas and hepatobiliary system. Furthermore, approximately 5-10% of the general population are Lewis antigen negative and lack the enzyme necessary to produce CA 19-9, leading to potentially false negative results. For these reasons, CA 19-9 cannot be used for screening purposes and the indication is mainly restricted to treatment monitoring.
The recent revolution in genomic, transcriptomic, proteomic and metabolomic technologies have contributed to the discovery of thousands of potential biomarker candidates for pancreatic cancer. These biomarkers are measurable in blood as liquid biopsies and have been evaluated either as single markers or as multimarker combinations, including protein panels5, 6, metabolites7, autoantibodies to tumour antigens8, 9, exosomes10, microRNAs11, nucleosomes12, circulating tumour cells13 and circulating tumour DNA14-16. Despite initial promise, no investigational biomarker has yet entered routine clinical practice. The question remains: Why do so many initially promising biomarkers fail to reach the clinic?
Figure 2. Biomarkers
On the surface, identifying and validating a biomarker candidate seems like an easy task. However, the reality is much more challenging and biomarker studies must be carefully planned. There are 5 phases to any biomarker development project directed at early detection17, including (1) preclinical exploratory studies, (2) clinical assay and validation studies, (3) retrospective longitudinal studies, (4) prospective screening studies and (5) cancer control studies. Most biomarker candidates fail in the earliest phases and the reasons may be multifactorial.
Lacking a clinically relevant question that biomarkers should address is a major failure factor18. It is not enough to distinguishing pancreatic cancer against healthy individuals. The biomarkers should also be able to differentiate between confounding factors such as inflammatory conditions (pancreatitis) or biliary obstruction. Another limitation is the use of serum samples obtained at the time of pancreatic cancer diagnosis. Several research groups now make use of pre-diagnostic samples to develop biomarkers7, 19-21.
The inherent complex tumour biology and heterogeneity of pancreatic cancer may also partly explain the biomarker failures. Traditionally, pancreatic cancer has been seen as a single disease entity, but recent genomic and transcriptomic studies have identified molecular subtypes of pancreatic cancer with distinct marker patterns and clinical behaviour22.
The objective of any early detection test must be to detect small, early stage tumours. The assay used to quantify the biomarkers of interest must therefore overcome the challenge of detecting miniscule amounts of circulating markers. Although imaging may be able to detect tumours that are less than 1 cm in size, it is still unclear if a very small tumour sheds enough molecules that can be quantified in blood with current technology23-26.
All biomarker assays struggle with specificity issues. This is especially a problem when looking for circulating tumour DNA. Some mutations can be misleading. Although KRAS mutations can be found in about 90% of pancreatic cancers, they are also prevalent in other malignant conditions, as well as pancreatic cysts that do not progress to cancer. It has been suggested to use a combination of circulating tumour DNA and protein markers to overcome this problem14.
Many biomarkers are also hampered by false discoveries and inability to reproduce data in external samples or by external laboratories. Reasons include poor study design, subpar samples, technical errors, bioinformatic limitations (e.g. overfitting) and bias27.
To enable earlier detection of pancreatic cancer, it is not enough to have an accurate biomarker test. The test must also be applied to the right population. Due to the low incidence of pancreatic cancer (approximately 13 per 100,000), the positive predictive value of the test is affected, which may lead to overdiagnosis. Therefore, it is well agreed that an early detection test should be deployed in a population of high-risk individuals. The most established risk group comprises individuals with a family history of pancreatic cancer or certain genetic mutations (e.g. CDKN2A, PRSS1, STK11, BRCA1, BRCA2, PALB2). These individuals have 2-132 times increased risk of developing pancreatic cancer28. Individuals with new onset diabetes mellitus after 50 years of age is another risk group, which has been found to have up to 8 times increased risk of developing pancreatic cancer within 3 years of diabetes diagnosis29. Some also suggest that a pancreatic cancer screening test should be applied in individuals with concerning, but non-specific symptoms, such as jaundice, abdominal pain, unexplained weight loss or fatigue, or individuals with cystic tumours of the pancreas (e.g. IPMN, MCN)30. In the future, artificial intelligence and deep learning algorithms may make use of personal health data to identify high-risk individuals that should be tested for pancreatic cancer31.
Figure 3. Artificial intelligence.
The road to new pancreatic cancer biomarkers is fraught with difficulties, but it also provides an opportunity to make a real difference. Pancreatic cancer is generally underfunded relative to its burden on society32 and long-term sustainable funding is essential for increasing research success rates. Biomarker development requires an interdisciplinary approach encompassing academia, health care, industry, regulators, government, philanthropic organizations and most importantly involvement of patients. A unified solution from initial biomarker discovery to final clinical implementation has for long been lacking. It is therefore encouraging that holistic roadmaps and guidelines have been published33-35. As a final thought, it is of minor importance which advanced technology that is used to discover a biomarker. In the end, the true value of a new biomarker will come down to clinical utility and simple measures such as sensitivity, specificity, and positive and negative predictive values.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin 2022;72:7-33.
Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin Pharmacol Ther 2001;69:89-95.
Koprowski H, Steplewski Z, Mitchell K, Herlyn M, Herlyn D, Fuhrer P. Colorectal carcinoma antigens detected by hybridoma antibodies. Somatic Cell Genet 1979;5:957-971.
Ballehaninna UK, Chamberlain RS. The clinical utility of serum CA 19-9 in the diagnosis, prognosis and management of pancreatic adenocarcinoma: An evidence based appraisal. J Gastrointest Oncol 2012;3:105-119.
Brand RE, Persson J, Bratlie SO, Chung DC, Katona BW, Carrato A, et al. Detection of Early-Stage Pancreatic Ductal Adenocarcinoma From Blood Samples: Results of a Multiplex Biomarker Signature Validation Study. Clin Transl Gastroenterol 2022;13:e00468.
Lindgaard SC, Sztupinszki Z, Maag E, Chen IM, Johansen AZ, Jensen BV, et al. Circulating Protein Biomarkers for Use in Pancreatic Ductal Adenocarcinoma Identification. Clin Cancer Res 2021;27:2592-2603.
Mayers JR, Wu C, Clish CB, Kraft P, Torrence ME, Fiske BP, et al. Elevation of circulating branched-chain amino acids is an early event in human pancreatic adenocarcinoma development. Nat Med 2014;20:1193-1198.
Bracci PM, Zhou M, Young S, Wiemels J. Serum autoantibodies to pancreatic cancer antigens as biomarkers of pancreatic cancer in a San Francisco Bay Area case-control study. Cancer 2012;118:5384-5394.
Capello M, Cappello P, Linty FC, Chiarle R, Sperduti I, Novarino A, et al. Autoantibodies to Ezrin are an early sign of pancreatic cancer in humans and in genetically engineered mouse models. J Hematol Oncol 2013;6:67.
Melo SA, Luecke LB, Kahlert C, Fernandez AF, Gammon ST, Kaye J, et al. Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature 2015;523:177-182.
Johansen JS, Calatayud D, Albieri V, Schultz NA, Dehlendorff C, Werner J, et al. The potential diagnostic value of serum microRNA signature in patients with pancreatic cancer. Int J Cancer 2016;139:2312-2324.
Bauden M, Pamart D, Ansari D, Herzog M, Eccleston M, Micallef J, et al. Circulating nucleosomes as epigenetic biomarkers in pancreatic cancer. Clin Epigenetics 2015;7:106.
Chen J, Wang H, Zhou L, Liu Z, Tan X. A combination of circulating tumor cells and CA199 improves the diagnosis of pancreatic cancer. J Clin Lab Anal 2022e24341.
Cohen JD, Li L, Wang Y, Thoburn C, Afsari B, Danilova L, et al. Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science 2018;359:926-930.
Guler GD, Ning Y, Ku CJ, Phillips T, McCarthy E, Ellison CK, et al. Detection of early stage pancreatic cancer using 5-hydroxymethylcytosine signatures in circulating cell free DNA. Nat Commun 2020;11:5270.
Klein EA, Richards D, Cohn A, Tummala M, Lapham R, Cosgrove D, et al. Clinical validation of a targeted methylation-based multi-cancer early detection test using an independent validation set. Ann Oncol 2021;32:1167-1177.
Pepe MS, Etzioni R, Feng Z, Potter JD, Thompson ML, Thornquist M, et al. Phases of biomarker development for early detection of cancer. J Natl Cancer Inst 2001;93:1054-1061.
Bender E. Will a test to detect early pancreatic cancer ever be possible? Nature 2020;579:S12-S13.
Nolen BM, Brand RE, Prosser D, Velikokhatnaya L, Allen PJ, Zeh HJ, et al. Prediagnostic serum biomarkers as early detection tools for pancreatic cancer in a large prospective cohort study. PLoS One 2014;9:e94928.
Duell EJ, Lujan-Barroso L, Sala N, Deitz McElyea S, Overvad K, Tjonneland A, et al. Plasma microRNAs as biomarkers of pancreatic cancer risk in a prospective cohort study. Int J Cancer 2017;141:905-915.
Borgmastars E, Lundberg E, Ohlund D, Nystrom H, Franklin O, Lundin C, et al. Circulating Tissue Polypeptide-Specific Antigen in Pre-Diagnostic Pancreatic Cancer Samples. Cancers (Basel) 2021;13.
Collisson EA, Bailey P, Chang DK, Biankin AV. Molecular subtypes of pancreatic cancer. Nat Rev Gastroenterol Hepatol 2019;16:207-220.
Hori SS, Gambhir SS. Mathematical model identifies blood biomarker-based early cancer detection strategies and limitations. Sci Transl Med 2011;3:109ra116.
Hori SS, Lutz AM, Paulmurugan R, Gambhir SS. A Model-Based Personalized Cancer Screening Strategy for Detecting Early-Stage Tumors Using Blood-Borne Biomarkers. Cancer Res 2017;77:2570-2584.
Lutz AM, Willmann JK, Cochran FV, Ray P, Gambhir SS. Cancer screening: a mathematical model relating secreted blood biomarker levels to tumor sizes. PLoS Med 2008;5:e170.
Avanzini S, Kurtz DM, Chabon JJ, Moding EJ, Hori SS, Gambhir SS, et al. A mathematical model of ctDNA shedding predicts tumor detection size. Sci Adv 2020;6.
Diamandis EP. The failure of protein cancer biomarkers to reach the clinic: why, and what can be done to address the problem? BMC Med 2012;10:87.
Klatte DCF, Wallace MB, Löhr M, Bruno MJ, van Leerdam ME. Hereditary pancreatic cancer. Best Practice & Research Clinical Gastroenterology 2022; Available online 3 January.
Chari ST, Leibson CL, Rabe KG, Ransom J, de Andrade M, Petersen GM. Probability of pancreatic cancer following diabetes: a population-based study. Gastroenterology 2005;129:504-511.
Pereira SP, Oldfield L, Ney A, Hart PA, Keane MG, Pandol SJ, et al. Early detection of pancreatic cancer. Lancet Gastroenterol Hepatol 2020;5:698-710.
Muhammad W, Hart GR, Nartowt B, Farrell JJ, Johung K, Liang Y, et al. Pancreatic Cancer Prediction Through an Artificial Neural Network. Front Artif Intell 2019;2:2.
Kamath SD, Kircher SM, Benson AB. Comparison of Cancer Burden and Nonprofit Organization Funding Reveals Disparities in Funding Across Cancer Types. J Natl Compr Canc Netw 2019;17:849-854.
Young MR, Wagner PD, Ghosh S, Rinaudo JA, Baker SG, Zaret KS, et al. Validation of Biomarkers for Early Detection of Pancreatic Cancer: Summary of The Alliance of Pancreatic Cancer Consortia for Biomarkers for Early Detection Workshop. Pancreas 2018;47:135-141.
Liu Y, Kaur S, Huang Y, Fahrmann JF, Rinaudo JA, Hanash SM, et al. Biomarkers and Strategy to Detect Preinvasive and Early Pancreatic Cancer: State of the Field and the Impact of the EDRN. Cancer Epidemiol Biomarkers Prev 2020;29:2513-2523.
Kenner BJ, Chari ST, Maitra A, Srivastava S, Cleeter DF, Go VL, et al. Early Detection of Pancreatic Cancer-a Defined Future Using Lessons From Other Cancers: A White Paper. Pancreas 2016;45:1073-1079.





.png)





.jpg)




