Normal View Dyslexic View
Surgical digest
Image-guided ablation for liver tumours – an addition to the armamentarium of multidisciplinary oncological and surgical approaches
The treatment of primary and metastatic liver tumours by ablation is not new. Advances in guidance systems, image fusion and new concepts of 3D tumour localisation and treatment, together with the current concepts of computer-assisted surgery that augment the senses (image-guided surgery), cognition (artificial intelligence), and execution (robotics), have enabled a fundamental change in the concept of ablation and have shaped image-guided ablation, also called precision ablation or Ablation 2.01. These changes have improved accuracy and in many cases made the oncological outcomes of ablation equal to those of surgical resection.
Indications for ablation therapies in liver tumours
Treatment options for colorectal liver metastases (CRLM) and hepatocellular carcinoma (HCC) include a combination of surgery, local ablation, and chemotherapy. For HCC, ablation is part of the treatment guidelines, such as the Barcelona Clinic Liver Cancer (BCLC) classification2, 3 and the ESMO (European Society for Medical Oncology) clinical practice guidelines4. It is used for smaller tumours and in patients with advanced cirrhosis, where resection may be more difficult3, 5. Liver resection is considered the gold standard for CRLM6-8. Recently, however, the COLLISION Trial Group presented a treatment algorithm for patients with CRLM without extrahepatic disease, recommending ablation for deeply situated metastases and unresectable metastases smaller than 3 cm7.
Ablation modalities
Tumour ablation is defined as the direct application of chemical, electrical or thermal energies to a target volume with the aim of inducing cell death in all viable neoplastic cells9. The modalities can be thermal via generated heat: radiofrequency ablation (RFA), and microwave ablation (MWA), or via extreme cold: cryoablation. The non-thermal technique of irreversible electroporation (IRE) uses high-voltage electrical pulses to permanently disrupt cell membrane function and induce cell death and apoptosis10. In addition, histotripsy is the first non-invasive, non-ionising, non-thermal ultrasonic ablation method, which is currently undergoing preliminary trials.
For liver tumours, the most widely used ablation techniques are RFA and MWA. RFA creates protein denaturation, coagulative necrosis, and immediate cell death by alternating electrical current with oscillations generating frictional heat (frequencies of 3Hz-300GHz, tissue temperature 60-100°C)9, 10. MWA is based on electromagnetic energy (frequencies of 915-2450MHz, tissue temperature >100°C) that induces coagulative necrosis by agitating surrounding water molecules. Compared to RFA, MWA induces higher intratumoural temperatures, larger ablation volumes and shorter ablation times9, 10.
Ablation outcomes
The ablative approaches were initially an alternative to resection when the risk of surgery was too high. For liver tumours that can be ablated completely, they have increasingly become the primary treatment. This is mostly limited to tumours less than 3 cm in diameter, allowing an ablation margin of 5-10 mm. Over the last decades, both energy deposition and targetting precision have improved significantly.
Currently, four randomised studies have been performed that compare ablation and resection for HCC, all from China11-14. Three of these showed similar overall survival, whereas in the other one,14 liver resection was favourable.
The role of ablation for patients with CRLM is less studied. In a recent prospective quasi-randomised study, MWA was associated with lower morbidity and similar overall survival to resection within two years of initial treatment15. Most retrospective studies using methods to decrease selection bias are in agreement16-18.
If ablation can offer an overall survival similar to liver resection, the choice of treatment modality should focus on other risks and benefits. Ablation offers a 75% reduction in complications, a greatly reduced operating time that frees up surgical capacity for other procedures, a much shorter hospital stay (from seven to one day), and lower costs for healthcare providers15.
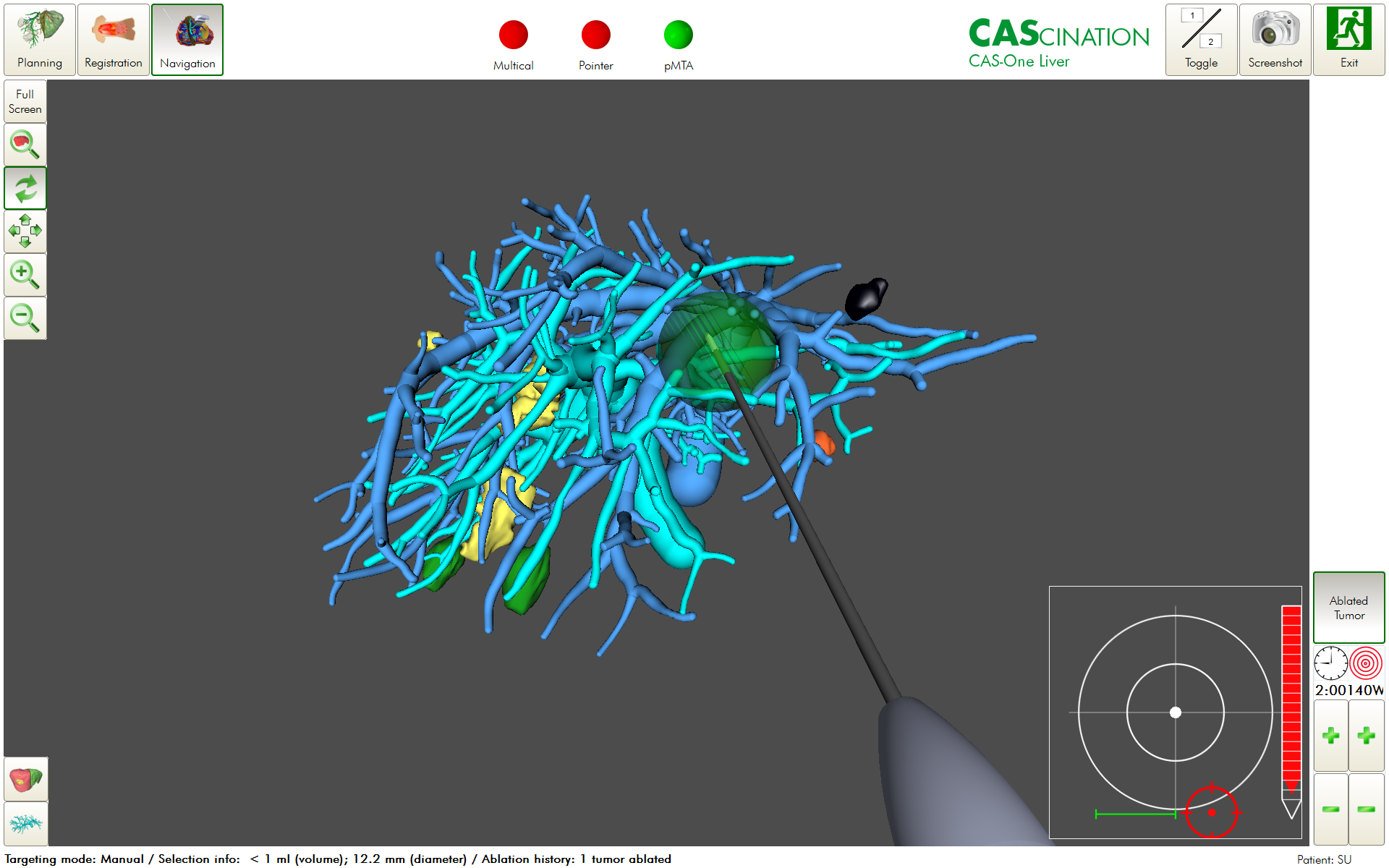
Recent studies using navigation guidance systems1 and precision ablation – organized by intraprocedural planning, execution and confirmation – have demonstrated low rates of local tumour progression (8,3%)19, similar to surgical resection (Figures 1 and 2). After resection or ablation, more than half of patients develop new tumours, need additional treatment, and thus benefit from minimally invasive and parenchyma-sparing approaches.
On the other hand, resection offers the opportunity to obtain large tissue samples for histopathological analysis and, in a future perspective, biomaterial for individualised tumour-targeted therapies.
Figure 1: Planning, Execution and Confirmation in Precision Ablation.
Figure 2: Augmented reality and electromagnetic navigation guidance system.
Interdisciplinary training
Ablation therapies require an optimal safety margin for local tumour control. The learning curve to achieve proficiency is usually long20. Therefore, dedicated training platforms are needed, including the use of complementary examination techniques such as intraoperative ultrasound (US) and computed tomography (Figures 3 and 4). Interpretation of spatial location, including depth and relationship to adjacent organs, is crucial for adequate planning, execution, and control, and to optimize oncological outcomes. There is limited information on training programmes and models to be used. However, skill improvement through training and following an adequate curriculum has been shown to improve the management of target tumours in ablative procedures significantly20, 21. Similarly, the use of advanced technology has the potential to equalise experiences, enabling trainees with little experience in tumour ablation to obtain expert-level results22.
Figure 3: Training in Laparoscopic Liver Ablation with laparoscopic ultrasound.
Figure 4: IHU Simulator training for Percutaneous Liver Ablation
Outlook and Perspectives
Local treatment of liver tumours should be minimally invasive, parenchyma-sparing, and sequential. These three characteristics are combined by tumour ablation. Precision ablation with navigation guidance systems or robotic assistance (fully or partially automated) enables oncological outcomes similar to surgical resection23. Additionally, it must be decided individually which procedure is most appropriate: either surgical resection or ablation, or a combination of both, sequentially or simultaneously. For ablation, the alternatives are percutaneous or laparoscopic.
Locoregional treatment such as ablation may also become a bridge to liver transplantation and major liver resections, or as a treatment option together with systemic chemotherapy in patients with unresectable CRLM10. For many patients with HCC, the waiting time for liver transplantation is long. Treating these patients with ablation whilst on the waiting may help to maintain their eligibility for transplant24.
The challenge today is to cross-train surgeons and radiologists in 3D image-guided ablation, with image fusion and the concept of computer-assisted surgery1. Finally, adaptation of the architectural structure of the operating theatre with hybrid operating rooms1 optimised for these procedures will improve and standardise the results of ablation therapies.
Funding information: This work was supported by French state funds managed within the “Plan Investissements d’Avenir” and by the ANR (reference ANR-10-IAHU-02).
References
Gimenez M, Gallix B, Costamagna G, Vauthey JN, Moche M, Wakabayashi G, Bale R, Swanstrom L, Futterer J, Geller D, Verde JM, Garcia Vazquez A, Boskoski I, Golse N, Muller-Stich B, Dallemagne B, Falkenberg M, Jonas S, Riediger C, Diana M, Kvarnstrom N, Odisio BC, Serra E, Overduin C, Palermo M, Mutter D, Perretta S, Pessaux P, Soler L, Hostettler A, Collins T, Cotin S, Kostrzewa M, Alzaga A, Smith M, Marescaux J. Definitions of Computer-Assisted Surgery and Intervention, Image-Guided Surgery and Intervention, Hybrid Operating Room, and Guidance Systems: Strasbourg International Consensus Study. Ann Surg Open 2020;1(2): e021.
Llovet JM, Di Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, Sherman M, Schwartz M, Lotze M, Talwalkar J, Gores GJ. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst 2008;100(10): 698-711.
BCLC-staging system. https://www.researchgate.net/figure/The-Barcelona-Clinic-Liver-Cancer-BCLC-staging-system-and-treatment-strategy-2011_fig1_258639064.
Vogel A, Cervantes A, Chau I, Daniele B, Llovet JM, Meyer T, Nault JC, Neumann U, Ricke J, Sangro B, Schirmacher P, Verslype C, Zech CJ, Arnold D, Martinelli E, Committee EG. Hepatocellular carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2018;29(Suppl 4): iv238-iv255.
Wang Y, Luo Q, Li Y, Deng S, Wei S, Li X. Radiofrequency ablation versus hepatic resection for small hepatocellular carcinomas: a meta-analysis of randomized and nonrandomized controlled trials. PLoS One 2014;9(1): e84484.
Di Martino M, Rompianesi G, Mora-Guzman I, Martin-Perez E, Montalti R, Troisi RI. Systematic review and meta-analysis of local ablative therapies for resectable colorectal liver metastases. Eur J Surg Oncol 2020;46(5): 772-781.
Nieuwenhuizen S, Puijk RS, van den Bemd B, Aldrighetti L, Arntz M, van den Boezem PB, Bruynzeel AME, Burgmans MC, de Cobelli F, Coolsen MME, Dejong CHC, Derks S, Diederik A, van Duijvendijk P, Eker HH, Engelsman AF, Erdmann JI, Fütterer JJ, Geboers B, Groot G, Haasbeek CJA, Janssen JJ, de Jong KP, Kater GM, Kazemier G, Kruimer JWH, Leclercq WKG, van der Leij C, Manusama ER, Meier MAJ, van der Meijs BB, Melenhorst M, Nielsen K, Nijkamp MW, Potters FH, Prevoo W, Rietema FJ, Ruarus AH, Ruiter SJS, Schouten EAC, Serafino GP, Sietses C, Swijnenburg RJ, Timmer FEF, Versteeg KS, Vink T, de Vries JJJ, de Wilt JHW, Zonderhuis BM, Scheffer HJ, van den Tol PMP, Meijerink MR. Resectability and Ablatability Criteria for the Treatment of Liver Only Colorectal Metastases: Multidisciplinary Consensus Document from the COLLISION Trial Group. Cancers (Basel) 2020;12(7).
Van Cutsem E, Cervantes A, Adam R, Sobrero A, Van Krieken JH, Aderka D, Aranda Aguilar E, Bardelli A, Benson A, Bodoky G, Ciardiello F, D'Hoore A, Diaz-Rubio E, Douillard JY, Ducreux M, Falcone A, Grothey A, Gruenberger T, Haustermans K, Heinemann V, Hoff P, Kohne CH, Labianca R, Laurent-Puig P, Ma B, Maughan T, Muro K, Normanno N, Osterlund P, Oyen WJ, Papamichael D, Pentheroudakis G, Pfeiffer P, Price TJ, Punt C, Ricke J, Roth A, Salazar R, Scheithauer W, Schmoll HJ, Tabernero J, Taieb J, Tejpar S, Wasan H, Yoshino T, Zaanan A, Arnold D. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol 2016;27(8): 1386-1422.
Uribe Rivera A, Ronchi A, Cilfone C, Guzmán Oviedo C, Cardoso G, Galliano L, Alvarez N, Franco N, Houghton E, Giménez M. Ablaciones percutáneas. In: Manual de técnicas en cirugía guiada por imágenes. Panamericana, 2023; 118-161. In press.
Lin YM, Paolucci I, Brock KK, Odisio BC. Image-Guided Ablation for Colorectal Liver Metastasis: Principles, Current Evidence, and the Path Forward. Cancers (Basel) 2021;13(16).
Ng KKC, Chok KSH, Chan ACY, Cheung TT, Wong TCL, Fung JYY, Yuen J, Poon RTP, Fan ST, Lo CM. Randomized clinical trial of hepatic resection versus radiofrequency ablation for early-stage hepatocellular carcinoma. Br J Surg 2017;104(13): 1775-1784.
Chen MS, Li JQ, Zheng Y, Guo RP, Liang HH, Zhang YQ, Lin XJ, Lau WY. A prospective randomized trial comparing percutaneous local ablative therapy and partial hepatectomy for small hepatocellular carcinoma. Ann Surg 2006;243(3): 321-328.
Feng K, Yan J, Li X, Xia F, Ma K, Wang S, Bie P, Dong J. A randomized controlled trial of radiofrequency ablation and surgical resection in the treatment of small hepatocellular carcinoma. J Hepatol 2012;57(4): 794-802.
Liu H, Wang ZG, Fu SY, Li AJ, Pan ZY, Zhou WP, Lau WY, Wu MC. Randomized clinical trial of chemoembolization plus radiofrequency ablation versus partial hepatectomy for hepatocellular carcinoma within the Milan criteria. Br J Surg 2016;103(4): 348-356.
Tinguely P, Laurell G, Enander A, Engstrand J, Freedman J. Ablation versus resection for resectable colorectal liver metastases - Health care related cost and survival analyses from a quasi-randomised study. Eur J Surg Oncol 2023;49(2): 416-425.
Uhlig J, Sellers CM, Stein SM, Kim HS. Radiofrequency ablation versus surgical resection of hepatocellular carcinoma: contemporary treatment trends and outcomes from the United States National Cancer Database. Eur Radiol 2019;29(5): 2679-2689.
Takayasu K, Arii S, Sakamoto M, Matsuyama Y, Kudo M, Kaneko S, Nakashima O, Kadoya M, Izumi N, Takayama T, Ku Y, Kumada T, Kubo S, Kokudo T, Hagiwara Y, Kokudo N, Liver Cancer Study Group of J. Impact of resection and ablation for single hypovascular hepatocellular carcinoma </=2 cm analysed with propensity score weighting. Liver Int 2018;38(3): 484-493.
Hocquelet A, Balageas P, Laurent C, Blanc JF, Frulio N, Salut C, Cassinotto C, Saric J, Possenti L, Bernard PH, Montaudon M, Trillaud H. Radiofrequency ablation versus surgical resection for hepatocellular carcinoma within the Milan criteria: A study of 281 Western patients. Int J Hyperthermia 2015;31(7): 749-757.
Schullian P, Johnston E, Laimer G, Putzer D, Eberle G, Amann A, Effenberger M, Maglione M, Freund MC, Loizides A, Bale R. Frequency and risk factors for major complications after stereotactic radiofrequency ablation of liver tumors in 1235 ablation sessions: a 15-year experience. Eur Radiol 2021;31(5): 3042-3052.
Diab K, Kochat S, McClintic J, Stevenson HL, Agle SC, Olino K, Tyler DS, Brown KM. Development of a Model for Training and Assessing Open Image-Guided Liver Tumor Ablation. J Surg Educ 2019;76(2): 554-559.
Garcia Vazquez A, Rodriguez-Luna MR, Verde J, Piantanida E, Alonci G, Palermo M, Serra E, De Cola L, Gimenez ME. Image-Guided Surgical Simulation in Minimally Invasive Liver Procedures: Development of a Liver Tumor Porcine Model Using a Multimodality Imaging Assessment. J Laparoendosc Adv Surg Tech A 2021;31(10): 1097-1103.
Davrieux CF, Gimenez ME, Gonzalez CA, Ancel A, Guinin M, Fahrer B, Serra E, Kwak JM, Marescaux J, Hostettler A. Mixed reality navigation system for ultrasound-guided percutaneous punctures: a pre-clinical evaluation. Surg Endosc 2020;34(1): 226-230.
Heerink WJ, Ruiter SJS, Pennings JP, Lansdorp B, Vliegenthart R, Oudkerk M, de Jong KP. Robotic versus Freehand Needle Positioning in CT-guided Ablation of Liver Tumors: A Randomized Controlled Trial. Radiology 2019;290(3): 826-832.
DuBay DA, Sandroussi C, Kachura JR, Ho CS, Beecroft JR, Vollmer CM, Ghanekar A, Guba M, Cattral MS, McGilvray ID, Grant DR, Greig PD. Radiofrequency ablation of hepatocellular carcinoma as a bridge to liver transplantation. HPB (Oxford) 2011;13(1): 24-32.





.png)





.jpg)




