Normal View Dyslexic View
Targeting intratumoral microbiome: the MONARCH Trial takes aim at anaerobes in rectal cancer
8 December 2025
Trials Lower GI
Locally advanced rectal cancer involves multimodality treatment that combines 5-fluorouracil (5-FU) based chemotherapy, chemoradiation and surgical resection. Total neoadjuvant therapy and radiation (TNT) has shown to induce complete tumor regression in up to 30-40% of patients1 and is associated with improved disease free survival.2 Furthermore, patients who attain a complete response to TNT, may be eligible for organ preserving strategies, offering them the potential to avoid radical resection and its attendant morbidity, including possible low anterior resection syndrome, bowel dysfunction, or permanent stoma formation.2-5 Despite its benefits, response to TNT remains markedly heterogeneous with more than half of patients not attaining a complete clinical response.6 The biological determinants underlying this variability remain an area many researchers are working to define. Identifying mechanisms that underlie variable treatment responses may enable novel strategies to enhance tumor regression and expand organ-preservation opportunities.
Emerging data from our group and others has suggested that the intertumoral microbiome may influence the variability in treatment response.7-9 Anaerobic bacteria, particularly Fusobacterium nucleatum8,10, have been associated with resistance to TNT and poor pathologic regression.11 Importantly, the intratumoral microbiome has shown to be dynamic9,12 and can be influenced by variables such as stress, diet, and therapies.13 Models have demonstrated that exposure to agents such as 5-FU or metronidazole can eradicate fusobacterium nucleatum and when eradicated patients had improved outcome.11,12,14,15
Building on these observations, we developed a phase II clinical trial to quantify intratumoral bacterial populations, particularly anaerobes and evaluate the impact of metronidazole administration on these tumoral bacterial populations. Metronidazole, an imidazole antibiotic, acts as a prodrug that is activated under anaerobic conditions, disrupts bacterial DNA synthesis ad metabolism.16,17 This trial aims to determine if selective depletion of anaerobes can be enhanced by the administration of metronidazole.
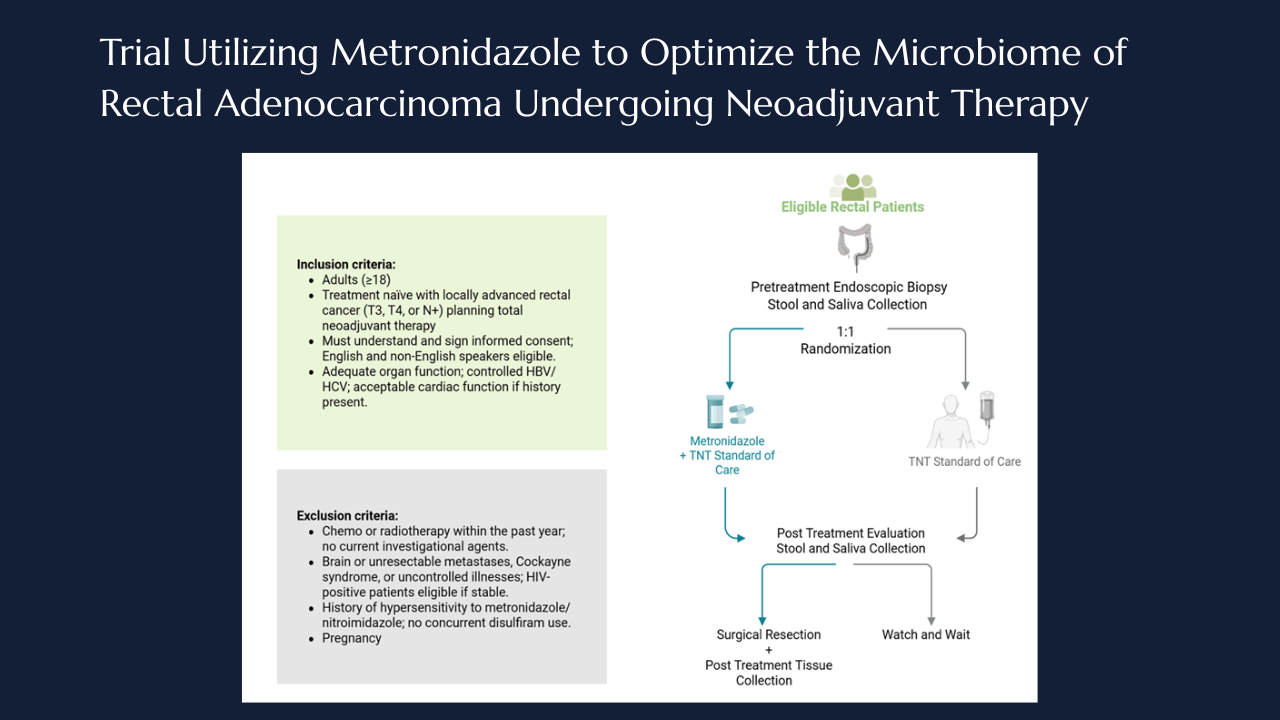
Study design:
Metronidazole Optimization for NeoAdjuvant Rectal adenocarcinoma Care and Health (MONARCH) is a prospective, randomized phase II clinical trial (ClinicalTrials.gov Identifier: NCT06569368) conducted at the University of Texas MD Anderson Cancer Center.
The trial enrolls patients who are treatment naïve and diagnosed with locally advanced rectal cancer (T3/4, N+), prior to initiation of TNT. Participants are randomized 1:1 to receive either standard of care neoadjuvant chemotherapy and/or chemoradiotherapy alone or standard therapy plus a 7-day course of oral metronidazole administered prior to or between their initial treatment cycles.
All participants undergo baseline sampling (timepoint 1), including endoscopic tumor biopsy as well as stool and saliva collection. Patients within the metronidazole arm the complete the 7 days of oral metronidazole, after which all participants proceed with TNT. Following the completion of TNT all the patients undergo standard restaging evaluation to determine clinical response, after which patients either undergo surgical resection or enter the watch and wait protocol. At the post treatment (timepoint 2), stool and saliva samples are recollected and tumor tissue is obtained at the time of surgery when applicable (Figure 1).
Figure 1.
Schematic of the MONARCH trial design. Created with BioRender.com.
The primary endpoint of the MONARCH trial is to quantify the ability of a 7-day course of metronidazole to reduce population of anaerobic bacteria within rectal tumors in patients who are undergoing neoadjuvant therapy. Secondary endpoints include assessment of pathologic response, recurrence free survival, overall survival, clinical complete response rate, and treatment related adverse events. In addition, exploratory analysis will evaluate changes in immune and microbial infiltrates within paired pre and post treatment tumor specimens to further elucidate the biologic effects of microbial modulation on the tumor microenvironment.
Goal enrollment is 78 patients with rectal cancer, randomized 1:1 to receive standard chemoradiotherapy or standard therapy plus a 7-day course of metronidazole. That will allow the study to be powered at 80% to detect differences in treatment response, accounting for an expected 30% complete clinical response rate suitable for organ preservation. Comprehensive clinical, microbiologic, and metagenomic analyses will evaluate the impact of metronidazole on the tumor microbiome and treatment response. Sequencing data will undergo quality control, human read removal, and bacterial classification to quantify anaerobic taxa. Clinical correlations will evaluate whether reductions in anaerobic bacteria align with improved tumor regression, recurrence-free survival, or overall survival, using Kaplan–Meier, log-rank, and Cox regression models.
The MONARCH trial is the first randomized study to test a metronidazole-based subtractive approach to reduce anaerobes and enhance TNT response in locally advanced rectal cancer. It will assess whether metronidazole decreases intratumoral anaerobes and improves complete clinical and pathologic response, recurrence-free, and overall survival. Surgeons play a critical role in the success of this trial. They can be involved by identifying and referring eligible patients with locally advanced rectal cancer and ensure that potential participants are informed of available clinical trial opportunities. Surgeons also can contribute by assisting with specimen acquisition in surgical tissue collection, which are integral to the trial's translational endpoints. Surgeons also can collaborate within the multidisciplinary care teams to inform others about these clinical trials and ensure integration of trials into treatment workflow.
Primary Contact:
Michael G. White, MD, MScDepartment of Colon and Rectal SurgeryThe University of Texas MD Anderson Cancer Center Houston, TX 77030Phone: 713-794-6940
Links and Key References:
ClinicalTrials.gov: NCT06569368 – Metronidazole to Optimize the Microbiome of Rectal Adenocarcinoma Undergoing Neoadjuvant TherapyMD Anderson Clinical Trial Page: Protocol 2023-0880
Funding declaration:
The study protocol and clinical trial are supported by an Early Career Award from the Colorectal Cancer Alliance, Society of University Surgeons Early Stage Investigator Award, and support from NIH Early Surgeon Scientist Program award as well as K12 (K12TR004908).
References
Kasi A, Abbasi S, Handa S, et al. Total Neoadjuvant Therapy vs Standard Therapy in Locally Advanced Rectal Cancer: A Systematic Review and Meta-analysis. JAMA Network Open. 2020;3(12):e2030097-e2030097. doi:10.1001/jamanetworkopen.2020.30097
Park IJ, You YN, Agarwal A, et al. Neoadjuvant treatment response as an early response indicator for patients with rectal cancer. J Clin Oncol. May 20 2012;30(15):1770-6. doi:10.1200/jco.2011.39.7901
Garcia-Aguilar J, Patil S, Gollub MJ, et al. Organ Preservation in Patients With Rectal Adenocarcinoma Treated With Total Neoadjuvant Therapy. J Clin Oncol. Aug 10 2022;40(23):2546-2556. doi:10.1200/jco.22.00032
Kupsch J, Jackisch T, Matzel KE, et al. Outcome of bowel function following anterior resection for rectal cancer-an analysis using the low anterior resection syndrome (LARS) score. Int J Colorectal Dis. Jun 2018;33(6):787-798. doi:10.1007/s00384-018-3006-x
Custers PA, van der Sande ME, Grotenhuis BA, et al. Long-term Quality of Life and Functional Outcome of Patients With Rectal Cancer Following a Watch-and-Wait Approach. JAMA Surgery. 2023;158(5):e230146-e230146. doi:10.1001/jamasurg.2023.0146
Ochiai K, Bhutiani N, Ikeda A, et al. Total Neoadjuvant Therapy for Rectal Cancer: Which Regimens to Use? Cancers (Basel). May 31 2024;16(11)doi:10.3390/cancers16112093
White MG, Damania A, Alshenaifi J, et al. Young-onset Rectal Cancer: Unique Tumoral Microbiome and Correlation With Response to Neoadjuvant Therapy. Ann Surg. Oct 1 2023;278(4):538-548. doi:10.1097/SLA.0000000000006015
Bullman S, Pedamallu CS, Sicinska E, et al. Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science. Dec 15 2017;358(6369):1443-1448. doi:10.1126/science.aal5240
Galeano Niño JL, Ponath F, Ajisafe VA, et al. Tumor-infiltrating bacteria disrupt cancer epithelial cell interactions and induce cell-cycle arrest. Cancer Cell. Oct 16 2025;doi:10.1016/j.ccell.2025.09.010
Zepeda-Rivera M, Minot SS, Bouzek H, et al. A distinct Fusobacterium nucleatum clade dominates the colorectal cancer niche. Nature. 2024/04/01 2024;628(8007):424-432. doi:10.1038/s41586-024-07182-w
Serna G, Ruiz-Pace F, Hernando J, et al. Fusobacterium nucleatum persistence and risk of recurrence after preoperative treatment in locally advanced rectal cancer. Ann Oncol. Oct 2020;31(10):1366-1375. doi:10.1016/j.annonc.2020.06.003
Galeano Niño JL, Wu H, LaCourse KD, et al. Effect of the intratumoral microbiota on spatial and cellular heterogeneity in cancer. Nature. Nov 2022;611(7937):810-817. doi:10.1038/s41586-022-05435-0
McQuade JL, Daniel CR, Helmink BA, Wargo JA. Modulating the microbiome to improve therapeutic response in cancer. The Lancet Oncology. 2019;20(2):e77-e91. doi:10.1016/S1470-2045(18)30952-5
Yu T, Guo F, Yu Y, et al. Fusobacterium nucleatum Promotes Chemoresistance to Colorectal Cancer by Modulating Autophagy. Cell. Jul 27 2017;170(3):548-563.e16. doi:10.1016/j.cell.2017.07.008
LaCourse KD, Zepeda-Rivera M, Kempchinsky AG, et al. The cancer chemotherapeutic 5-fluorouracil is a potent Fusobacterium nucleatum inhibitor and its activity is modified by intratumoral microbiota. Cell Rep. Nov 15 2022;41(7):111625. doi:10.1016/j.celrep.2022.111625
Dingsdag SA, Hunter N. Metronidazole: an update on metabolism, structure-cytotoxicity and resistance mechanisms. J Antimicrob Chemother. Feb 1 2018;73(2):265-279. doi:10.1093/jac/dkx351
Lofmark S, Edlund C, Nord CE. Metronidazole is still the drug of choice for treatment of anaerobic infections. Clin Infect Dis. Jan 1 2010;50 Suppl 1:S16-23. doi:10.1086/647939
Published on: 8 December 2025
Article ID: BJS-2025-0583






.png)





.jpg)




