Major digestive surgery not only carries clinical risks but also imposes a significant functional and emotional burden. In this context, prehabilitation emerges as a key strategy to optimize patient readiness before entering the operating room.
Study context
This analysis is based on a prospective cohort study conducted at a tertiary hospital, including 188 patients undergoing major elective gastrointestinal surgery. Of these, 94 completed a multimodal prehabilitation program, while 94 received standard preoperative care (Table 1). Outcomes were assessed using the validated QoR-15 questionnaire at baseline, discharge, and 30 days post-discharge, alongside clinical and functional recovery indicators (Figure 1).
Table 1
Preoperative clinic data and type of surgery by group
| Prehabilitation n = 94 | Control n = 94 | p value | |
| Age (years) | 71.47 ± 10.3 | 70.04 ± 12.7 | 0.666 |
| Gender (female) | 36 (38.3) | 50 (53.2) | 0.059 |
| ASA ≥ III | 68 (72.4) | 51 (54.3) | 0.029 |
| CHSA Frailty ≥ IV | 45 (47.9) | 36 (38.3) | 0.009 |
| Neoadjuvant therapy | 37 (39.4) | 18 (19.1) | 0.007 |
| Type of surgery | 0.321 | ||
| Colorectal resection | 34 (36.2) | 34 (36.2) | |
| Liver resection | 6 (6.4) | 7 (7.4) | |
| Bariatric | 2 (2.1) | 3 (3.2) | |
| Gastrectomy | 8 (8.5) | 7 (7.4) | |
| Esophagectomy | 19 (20.2) | 8 (8.5) | |
| Pancreatectomy | 8 (8.5) | 15 (16.0) | |
| HIPEC | 9 (9.6) | 13 (13.8) | |
| Other | 8 (8.5) | 7 (7.4) | |
| Surgical approach | 0.982 | ||
| Open | 21 (22.3) | 24 (25.5) | |
| Laparoscopic | 38 (40.4) | 36 (38.3) | |
| Robotic | 35 (37.2) | 34 (36.2) | |
| Surgical time (minutes) | 282.7 ± 149.6 | 282.6 ± 162.2 | 0.900 |
| Table 1. Continuous variables are reported as mean ± standard deviation or median and interquartile range depending on their distribution, and categorical variables as absolute and relative frequencies (n (%)). ASA: American Society of Anesthesiologists. CHSA: Canadian Health and Safety Assessment, (which classifies patients into nine levels based on their functional status and degree of dependency, ranging from “very fit” (1) to “terminally ill” (9). Patients scoring 4 or above can be considered vulnerable). HIPEC: Hyperthermic Intraperitoneal Chemotherapy. Other: complex abdominal wall surgery, supra-adrenalectomy. All included procedures were classified as major surgeries according to the criteria of the National Institute for Health and Care Excellence (NICE). p-values represent comparisons between the two cohorts. | |||
Figure 1
Sankey diagram illustrating the evolution of QoR-15 scores over time. Green bars represent patients with an excellent QoR-15 rating, yellow bars correspond to good ratings, orange bars to moderate ratings, and red bars to poor ratings. The diagram shows the evolution of the number of patients in each cohort from baseline (T0, before surgery) to discharge day (TD) and 30 days postoperatively (T30). Prehabilitation patients maintained a more stable distribution across categories over time, with fewer patients shifting toward worse QoR-15 categories at TD and a greater proportion returning to higher categories by T30, compared to the control cohort.
What was the impact on perceived recovery?
Beyond clinical benefits, prehabilitation improved the patient experience. Key findings include:
Quality of Recovery (QoR-15): Prehabilitation consistently improved scores at discharge and 30 days, helping patients return to baseline faster than controls (discharge: p < 0.001; 30 days: p < 0.001), (Table 2).
Functional Recovery: By 30 days, more prehabilitated patients had resumed all three self-identified essential activities compared with controls (p = 0.001).
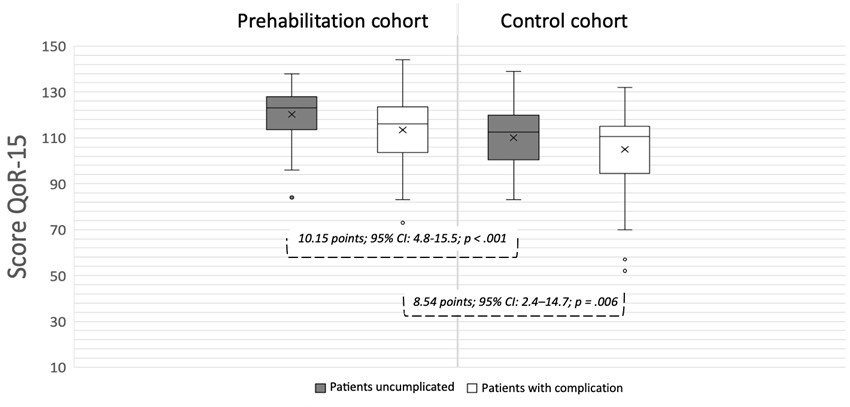
Complications: Prehabilitation was associated with fewer postoperative complications (p = 0.008),(Table 3), and even when complications occurred (Figure 2), patients reported better recovery than controls (p = 0.006), indicating benefits beyond simply reducing adverse events.
Table 2
QoR-15 scores at different time points
| Prehabilitation (n=94) | Control (n=94) | p value | |
| Baseline QoR-15 score (T0) | 129.5 ± 15.0 | 122.9 ± 17.0 | 0.003 |
| Baseline QoR-15 category | 0.001 | ||
| Excellent | 42 (45) | 18 (20) | |
| Good | 22 (23) | 44 (47) | |
| Moderate | 29 (31) | 26 (28) | |
| Poor | 1 (1) | 5 (5) | |
| Discharge QoR-15 score (TD) | 117.18 ± 14.0 | 106.83 ± 15.0 | < 0.001 |
| Discharge QoR-15 category | < 0.001 | ||
| Excellent | 4 (4) | 2 (2) | |
| Good | 35 (38) | 10 (11) | |
| Moderate | 52 (55) | 70 (74) | |
| Poor | 3 (3) | 12 (13) | |
| 30-day QoR-15 score (T30) | 128.24 ± 16.0 | 118.47 ± 14.0 | < 0.001 |
| 30-day QoR-15 category | < 0.001 | ||
| Excellent | 34 (37) | 8 (9) | |
| Good | 31 (34) | 42 (45) | |
| Moderate | 27 (29) | 40 (43) | |
| Poor | 0 | 3 (3) | |
| Table 2. QoR-15 categories were classified as: Excellent (≥ 136), Good (122–135), Moderate (90–121), and poor (< 90). p-values represent comparisons between the two cohorts. | |||
Table 3
Postoperative outcomes
| Prehabilitation (n = 94) | Control (n = 94) | p value | |
| Patients with no complications during admission | 49 (52) | 34 (36) | < 0.001 |
| Total number of complications | 0.98 ± 1.4 | 1.43 ± 1.4 | 0.008 |
| CCI | 11.86 ± 16 | 14.96 ± 15 | 0.290 |
| Length of hospital stay (days) | 8.72 ± 9 | 9.16 ± 12 | 0.622 |
| ICU admission | 45 (48) | 41 (44) | 0.526 |
| ICU* length of hospital stay (days) | 3 ± 4.6 | 2.46 ± 2.8 | 0.518 |
| Patients destination at hospital discharge | 0.003 | ||
| Discharge to home | 76 (82.6) | 67 (71.3) | |
| Discharge to home with support services | 10 (10.9) | 25 (28.5) | |
| Post discharge emergency visits | 16 (17) | 27 (29) | 0.071 |
| Post discharge re-admissions | 11 (12) | 14 (16) | 0.367 |
| Recovered selected activities at 30 days post discharge | 0.001 | ||
| 0 | 6 (6) | 11 (1) | |
| 1 | 9 (10) | 25 (30) | |
| 2 | 20 (23) | 28 (34) | |
| 3 | 57 (66) | 29 (35) | |
| Table 3. Continuous variables are reported as mean ± standard deviation or median and interquartile range depending on their distribution, and categorical variables as absolute and relative frequencies (n (%)). CCI: Comprehensive Complication Index;*ICU days: calculated only for patients admitted to the intensive care unit. | |||
Figure 2
QoR-15 scores at hospital discharge by cohort and complication status. Boxplots represent: prehabilitation without complications (mean 120.2 ± 11.1), prehabilitation with complications (113.5 ± 15.1), control without complications (110.0 ± 13.0), and control with complications (105.0 ± 15.9)
How was this achieved?
The program was multimodal, combining three pillars:
Supervised exercise training (aerobic and resistance, 2–3 sessions per week, tailored to baseline capacity, plus daily activity goals and breathing exercises for high pulmonary risk).
Nutritional optimization, including systematic assessment, dietary counseling, and supplementation for malnourished patients.
Psychological support, with HAD screening and individualized follow-up for anxiety or depression.
Adherence to the exercise component was remarkable: patients completed 86% of scheduled sessions over an average of 4.5 weeks, demonstrating feasibility in real-world practice. This commitment translated into preoperative functional gains and, ultimately, a more positive recovery experience. Notably, lower-limb strength measured by the 30-Second Sit-to-Stand (30STS) test remained improved at 30 days post-discharge (+1.1 repetitions; p = 0.002).
The study was conducted under pragmatic conditions, reflecting routine implementation of prehabilitation in a tertiary hospital. Importantly, the program targeted clinically complex patients, including those with ASA III–IV and higher frailty, underscoring its applicability in vulnerable populations.
Implications for surgical practice
For surgeons, success should not be defined solely by survival or absence of complications. The ultimate goal is for patients to regain functionality and well-being as quickly as possible. This study provides evidence that prehabilitation:
Optimizes physical and psychological readiness, translating into better perceived recovery.
Enhances autonomy, reducing dependency after discharge.
Complements ERAS protocols, adding value in terms of patient-centered outcomes.
In an era prioritizing efficiency and humanization, integrating prehabilitation into routine surgical care can make a meaningful difference.
Reflections and future directions
This study offers real-world evidence that multimodal prehabilitation is feasible and effective even in high-risk patients. While its observational, single-center design limits generalizability, the magnitude of benefit and adjusted analyses confirm its clinical relevance.
Next steps include:
Validating these findings in multicenter studies across diverse settings, to strengthen external validity.
Developing strategies for broader implementation, ensuring equitable access for all eligible patients.
Exploring long-term outcomes, including quality of life and functional independence beyond 30 days postoperatively.
Take-home message
Surgery does not end in the operating room. Preparing patients to reclaim their lives—not just survive the procedure—is the true objective. Prehabilitation stands out as a strategy that improves not only clinical outcomes but also patient experience and quality of life. Are we ready to take this step?
References
1.Dana F, González-Colom R, Tena B, Capitán D, Momblan D, Campero B, et al. Impact of prehabilitation on patient-perceived quality of recovery after surgery: prospective cohort study, BJS Open. doi: https://doi.org/10.1093/bjsopen/zraf156
2.Stark PA, Myles PS, Burke JA. Development and psychometric evaluation of a postoperative quality of recovery score: the QoR-15. Anesthesiology. 2013;118:1332–1340. doi:10.1097/ALN.0b013e318289b84b.
3.Morales-Ariza V, Loaiza-Aldeán Y, de Miguel M, Peña-Navarro M, Martínez-Silva O, González-Tallada A, et al. Validation and cross-cultural adaptation of the Postoperative Quality of Recovery 15 (QoR-15) questionnaire for Spanish-speaking patients: a prospective cohort study. Am J Surg. 2022;S0002-9610(22)00719-X. doi:10.1016/j.amjsurg.2022.11.009.
4.Myles PS, Myles D. An updated minimal clinically important difference for the QoR-15 scale. Anesthesiology. 2021;135:934–935. doi:10.1097/ALN.0000000000003977.
5.Fleurent-Gregoire C, Burgess N, McIsaac DI, Chevalier S, Fiore JF Jr, Carli F, et al. Towards a common definition of surgical prehabilitation: a scoping review of randomised trials. Br J Anaesth. 2024;133:305–315. doi:10.1016/j.bja.2024.02.035.
6.Barberan-García A, Ubré M, Roca J, Lacy AM, Burgos F, Risco R, et al. Personalised prehabilitation in high-risk patients undergoing elective major abdominal surgery: a randomized blinded controlled trial. Ann Surg. 2018;267:50–56. doi:10.1097/SLA.0000000000002293.
7.Molenaar CJL, Minnella EM, Coca-Martinez M, Ten Cate DWG, Regis M, Awasthi R, et al. Effect of Multimodal Prehabilitation on Reducing Postoperative Complications and Enhancing Functional Capacity Following Colorectal Cancer Surgery: The PREHAB Randomized Clinical Trial. JAMA Surg. 2023;158:572–581. doi:10.1001/jamasurg.2023.0198.
8.Risco R, González-Colom R, Montané-Muntané M, Cano I, Vela E, Sebio R, et al. Actionable factors fostering health valUe generation and scalability of prehabilitation: a prospective cohort study. Ann Surg. 2022 Aug 15. doi:10.1097/SLA.0000000000005662.
9.Ten Cate DWG, Molenaar CJL, Garcia RS, Bojesen RD, Tahasildar BLR, Jansen L, et al. Multimodal prehabilitation in elective oncological colorectal surgery enhances postoperative functional recovery: A secondary analysis of the PREHAB randomized clinical trial. Eur J Surg Oncol. 2024;50:108270. doi:10.1016/j.ejso.2024.108270.
10.Ljungqvist O, Scott M, Fearon KC. Enhanced recovery after surgery: a review. JAMA Surg. 2017;152:292–298. doi:10.1001/jamasurg.2016.4952.
11.Dana F, Sebio-García R, Tena B, Sisó M, Vega F, et al. Perioperative nursing as the guiding thread of a prehabilitation program. Cancers (Basel). 2022;14:5376. doi:10.3390/cancers14215376.






.png)





.jpg)




