Normal View Dyslexic View
Personalized neoadjuvant immunotherapy for stage III malignant melanoma: notes on the PRADO study
Background
While significant changes have occurred in the management of microscopic stage III disease due to the data from MSLT-21, the current standard of care for macroscopic stage III nodal malignant melanoma consists of initial surgical treatment with therapeutic lymph node dissection (TLND), followed by consideration for adjuvant therapy consisting of either anti-PD-1 monotherapy2 or BRAF/MEK inhibitors3. This results in improved relapse-free survival, but recurrence is still observed in almost half of the patients within 3-5 years4-6. Preclinical trials7-9 and emerging clinical data10 suggest that neoadjuvant immune checkpoint inhibition may have clinical benefit over adjuvant approaches. Given increasing enthusiasm for adjuvant and neoadjuvant approaches, the OpACIN (NCT02437279, phase I) and OpACIN-neo (NCT02977052, phase II) studies were established to investigate the safety and efficacy of neoadjuvant treatment with immune checkpoint inhibitors (ICI) combination, and to establish optimal dosing regimens to maximize clinical benefit while minimizing toxicity in patients with stage III melanoma.
The OpACIN and OpACIN-neo trials
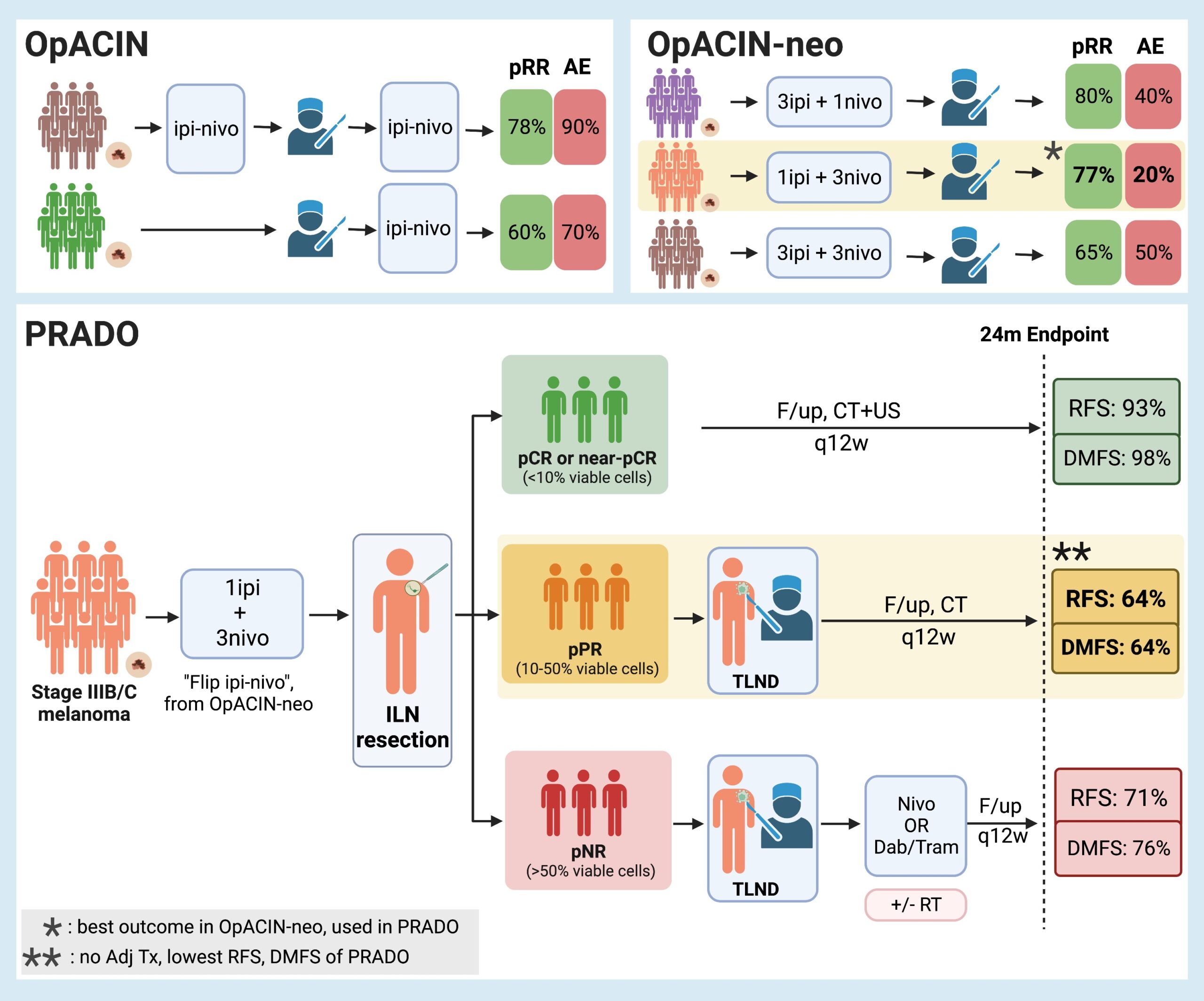
The OpACIN study, a two-arm phase Ib trial, evaluated the efficacy of the combination of ipilimumab (anti-cytotoxic T-cell lymphocyte antibody – anti-CTLA) and nivolumab (anti-programmed cell death protein 1 – anti-PD-1) in an adjuvant or split neoadjuvant-adjuvant fashion for a limited number of patients (10 patients in each arm). Primary data from that study in 201811 demonstrated the remarkable efficacy of the neoadjuvant-adjuvant combinations, with a pathological response rate (pRR) of 78% after neoadjuvant therapy. However, toxicity was a significant concern, as grade 3 and 4 adverse events related to the immunotherapy affected 90% of the participants. For comparison, the pRR and adverse events (AE) in the adjuvant arm of the study were 60% and 70%, respectively. These results are comparable with the results of other trials12.
The subsequent phase II OpACIN-neo trial further investigated the safety and efficacy of neoadjuvant combinations of ipilimumab and nivolumab for stage III melanoma at varying doses. The randomized multicenter study comprised three arms with different neoadjuvant combination doses (Arm A: 2 courses of ipilimumab 3mg/kg and nivolumab of 1mg/kg; Arm B: 2 courses of ipilimumab 1mg/kg and nivolumab 3mg/kg; Arm C: 2 courses of ipilimumab 3mg/kg followed by 2 courses of nivolumab 3mg/kg). The 86 patients enrolled in the study were randomized in a 1:1:1 fashion, with almost 30 patients per arm (A:30, B:29, C:24). At a follow-up of 24 months, the AE related to the immunotherapy course and pRR were: Arm A: 40% irAE and 80% pRR, Arm B: 20% irAE and 77% pRR, and Arm C: 50% irAE and 65% pRR. Based on this the Arm B, often termed flip-dose Ipi/Nivo (IPI 1mg/kg, NIVO 3mg/kg), was identified as the optimal dosing regimen since it demonstrated the lowest toxicity (20%) with an equivalent pathological response rate (77%).
The MeMaLoc substudy
Within the OpACIN-neo study, 12 patients were enrolled in the MeMaLoc pilot trial (Magnetic Seed Localization for Melanoma, NL58293.031.16), a substudy examining whether the pathological response of the index lymph node (ILN) to treatment could accurately predict the response of the total lymph node basin. A magnetic seed was placed in the ILN in a similar fashion to breast surgery13, and the data demonstrated 100% concordance between the magnetically marked and resected ILN with the entire basin after neoadjuvant therapy (12/12 cases)14. This proved that the localization and resection of the index node is safe, feasible, and reliable, thus paving the way for patients with a complete response potentially to avoid a TLND in the future.
The PRADO extension cohort of OpACIN-neo
The extension cohort of the OpACIN-neo trial called PRADO constitutes a multicenter cohort study that assessed the effect of personalized surgical and adjuvant treatment based on the response of patients with stage III melanoma to the neoadjuvant therapy after 2 cycles of “flip-dose Ipi/Nivo”, i.e. ipilimumab 1mg/kg and nivolumab 3mg/kg (the most successful arm of the OpACIN-neo trial). The response to the neoadjuvant immune checkpoint inhibitor (ICI) combination was measured through ILN assessment with the use of a magnetic seed (52%) (in the fashion of the MeMaLoc study), a nitinol marker (34%), a radioactive I125 seed (9%), or a hydrogel marker (4%).
If the ILN had a major pathological response (MPR), i.e. a complete response (CR) or near CR (nCR), with <10% viable tumour cells remaining, TLDN was omitted along with any further adjuvant immunotherapy or radiotherapy. If the ILN showed a pathological partial response (PR), with 10-50% of viable tumour cells left, then TLDN followed the neoadjuvant therapy, but no further adjuvant immune or radiotherapy was offered. Lastly, in the case of non-response (NR, >50% viable tumour cells left in the ILN), TLDN took place, followed by standard-of-care adjuvant therapy consisting of either nivolumab (for BRAF wild-type tumours) or the BRAF/MEK inhibitors dabrafenib and trametinib (for BRAFV600E/K-mutated tumours), along with the possible addition of local radiotherapy.
A total of 99 patients who met the RECIST 1.115 criteria were enrolled. After 6 weeks of neoadjuvant therapy, 62% achieved MPR (49% CR, 12% nCR) and thus avoided TLND, 11% achieved PR and thus underwent only TLND, and 21% had NR and underwent TLND. Of the 21 patients with NR who underwent TLDN, only 17 followed with adjuvant therapy ( 7 of them with nivolumab and 10 with a combination of the BRAF/MEK inhibitors). Furthermore, 8 out of 17 patients received additional local radiotherapy.
The first outcome of the study was the evaluation of the morbidity of TLND. Avoiding TLDN resulted in a significant reduction of morbidity as there was a significantly lower rate of surgery-related adverse events (based on the CTCAE v4 criteria) in patients who underwent ILN resection alone compared to those with ILN and TLND (46% versus 84%, p<0.001). There was the same trend shown with the Clavien-Dindo classification (52% vs 93%, p<0.001). Similarly, the HRQoL rates were significantly increased in the group that omitted the TLND.
The next outcomes of the study were the overall survival (OS), relapse-free survival (RFS), and distant-metastasis-free survival (DMFS) rates of the participants. At a 24-month endpoint, the combined OS for patients was 95%, with RFS rates for the patients with MPR being 93%, PR of 64%, and NR of 71% respectively, with similar DMFS rates observed (MPR: 98%, PR: 64%, and NR: 64%). It was unexpected that the RFS rates for those with a PR was similar to that with neoaduvant pathological NR. However the PR patients had a TLND, but did not (per study protocol) receive adjuvant therapy.
In conclusion, the PRADO study, based on the promising outcomes of the OpACIN and OpACIN-neo, and using the pioneering tools from the MeMaLoc study, made some interesting observations, which can be summarized as follows:
It supported the efficacy and safety of the OpACIN-neo trial’s most favourable neoadjuvant treatment arm (ipilimumab 1mg/kg and nivolumab 3mg/kg).
The study validated the data supporting that the ILN response can be an accurate representation of the entire tumour basin.
The study made a significant step towards precision medicine of stage III melanoma, as it showed that de-escalation is possible in patients with MPR, whereas in patients with PR or NR, escalation might improve their outcome.
While the encouraging outcomes of those with a major pathological response with focused nodal surgery support consideration for less invasive approaches to surgery after neoadjuvant therapy, the integration of surgery and neoadjuvant/adjuvant choices should be considered thoughtfully. The disappointing outcomes of the PR group who received TLND but did not have additional adjuvant therapy suggests that de-escalation of surgery without adjuvant therapy may be a less optimal approach, since their outcomes paralleled that of the pathological NR group. Future efforts at patient-specific therapy decisions should consider adjuvant therapy for both PR and NR groups. However, this does not dampen the importance of this study in validating the importance of a major pathological response as an opportunity to de-escalate surgery.
Overall, neoadjuvant therapy, ILN assessment and consequent response-directed treatment are significant added tools that could potentially be added to the oncological treatment of stage III melanoma and achieve even better outcomes. Further clinical trials are needed to examine this in more detail, expanding and further illuminating this thought-provoking and noteworthy topic.
References
Faries MB, Thompson JF, Cochran AJ, Andtbacka RH, Mozzillo N, Zager JS et al. Completion Dissection or Observation for Sentinel-Node Metastasis in Melanoma. New England Journal of Medicine 2017;376(23): 2211-2222.
Weber J, Mandala M, Del Vecchio M, Gogas HJ, Arance AM, Cowey CL et al. Adjuvant Nivolumab versus Ipilimumab in Resected Stage III or IV Melanoma. N Engl J Med 2017;377(19): 1824-1835.
Long GV, Hauschild A, Santinami M, Atkinson V, Mandalà M, Chiarion-Sileni V et al. Adjuvant Dabrafenib plus Trametinib in Stage III BRAF-Mutated Melanoma. N Engl J Med 2017;377(19): 1813-1823.
Dummer R, Brase JC, Garrett J, Campbell CD, Gasal E, Squires M et al. Adjuvant dabrafenib plus trametinib versus placebo in patients with resected, BRAF(V600)-mutant, stage III melanoma (COMBI-AD): exploratory biomarker analyses from a randomised, phase 3 trial. Lancet Oncol 2020;21(3): 358-372.
Eggermont AM, Blank CU, Mandalà M, Long GV, Atkinson V, Dalle S et al. Pembrolizumab versus placebo after complete resection of high-risk stage III melanoma: New recurrence-free survival results from the EORTC 1325-MG/Keynote 054 double-blinded phase III trial at three-year median follow-up. Journal of Clinical Oncology 2020;38(15_suppl): 10000-10000.
Ascierto PA, Del Vecchio M, Mandalá M, Gogas H, Arance AM, Dalle S et al. Adjuvant nivolumab versus ipilimumab in resected stage IIIB-C and stage IV melanoma (CheckMate 238): 4-year results from a multicentre, double-blind, randomised, controlled, phase 3 trial. Lancet Oncol 2020;21(11): 1465-1477.
Liu J, Blake SJ, Yong MC, Harjunpää H, Ngiow SF, Takeda K et al. Improved Efficacy of Neoadjuvant Compared to Adjuvant Immunotherapy to Eradicate Metastatic Disease. Cancer Discov 2016;6(12): 1382-1399.
Blank CU, Rozeman EA, Fanchi LF, Sikorska K, van de Wiel B, Kvistborg P et al. Neoadjuvant versus adjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma. Nature Medicine 2018;24(11): 1655-1661.
O'Donnell JS, Hoefsmit EP, Smyth MJ, Blank CU, Teng MWL. The Promise of Neoadjuvant Immunotherapy and Surgery for Cancer Treatment. Clin Cancer Res 2019;25(19): 5743-5751.
S. Patel, M. Othus, V. Prieto, M. Lowe, E. Buchbinder, Y. Chen, J et al. Neoadjvuant versus adjuvant pembrolizumab for resected stage III-IV melanoma (SWOG S1801). In: ESMO Congress 2022. Paris, France: Annals of Oncology; 2022. p. S808-S869.
Rozeman EA, Hoefsmit EP, Reijers ILM, Saw RPM, Versluis JM, Krijgsman O et al. Survival and biomarker analyses from the OpACIN-neo and OpACIN neoadjuvant immunotherapy trials in stage III melanoma. Nature Medicine 2021;27(2): 256-263.
Amaria RN, Reddy SM, Tawbi HA, Davies MA, Ross MI, Glitza IC et al. Neoadjuvant immune checkpoint blockade in high-risk resectable melanoma. Nat Med 2018;24(11): 1649-1654.
Schermers B, van der Hage JA, Loo CE, Vrancken Peeters MTFD, Winter-Warnars HAO, van Duijnhoven F et al. Feasibility of magnetic marker localisation for non-palpable breast cancer. The Breast 2017;33: 50-56.
Schermers B, Franke V, Rozeman EA, van de Wiel BA, Bruining A, Wouters MW et al. Surgical removal of the index node marked using magnetic seed localization to assess response to neoadjuvant immunotherapy in patients with stage III melanoma. Br J Surg 2019;106(5): 519-522.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45(2): 228-247.





.png)





.jpg)




