BJS Academy>Surgical science>Discussion of: decel...
Discussion of: decellularized adipose matrices can alleviate radiation-induced skin fibrosis; adv wound care (New Rochelle).
Kanad Ghosh, MD1; Hannes Prescher, MD1; Summer E. Hanson, MD, PhD, FACS1
1Section of Plastic and Reconstructive Surgery, Department of Surgery, University of Chicago Medicine and Biological Sciences, Chicago, IL, 60637
Related articles
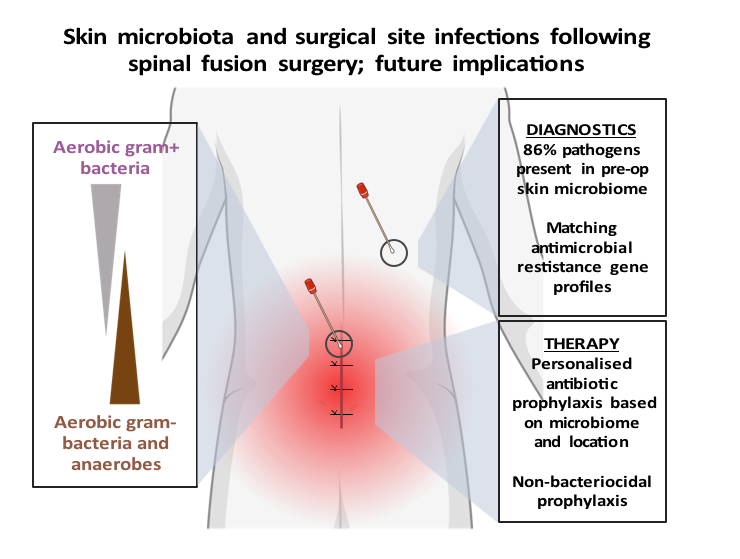
Review of: Contribution of the patient microbiome to surgical site infection and antibiotic prophylaxis failure in spine surgery
F.F. van den Berg1, M.A. Boermeester2
1Department of Medical Microbiology, Amsterdam UMC, Amsterdam, the Netherlands
2Department of Surgery, Amsterdam UMC, Amsterdam, the Netherlands
Article review Long, D. R. et al. Contribution of the patient microbiome to surgical site infection and antibiotic prophylaxis failure in spine surgery. Sci. Transl. Med.16, eadk8222 (2024). https://www.science.org/doi/10.1126/scitranslmed.adk8222
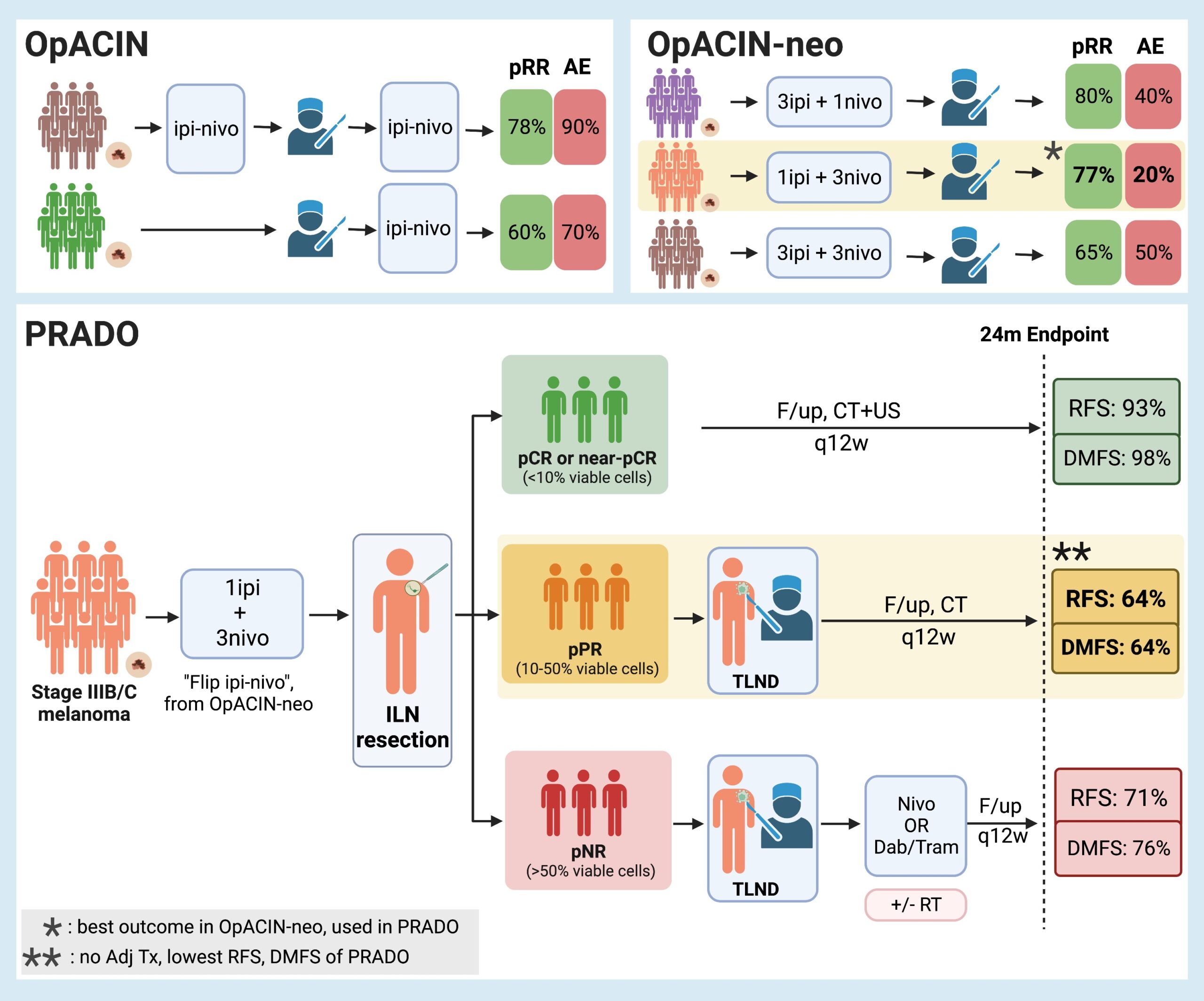
Personalized neoadjuvant immunotherapy for stage III malignant melanoma: notes on the PRADO study
1Aikaterini Dedeilia, M.D, 2Genevieve Boland, MD, PhD
1Postdoctoral Research Fellow, Surgical Oncology Research Laboratories; Department of Surgery, Massachusetts General Hospital; Research Fellow in Surgery, Harvard Medical School.
2Vice Chair of Research, Department of Surgery; Section Head, Melanoma/Sarcoma Surgery; Surgical Director, Termeer Center for Targeted Therapies; Director, Surgical Oncology Research Laboratories, Massachusetts General Hospital; Associate Professor of Surgery, Harvard Medical School.
Background While significant changes have occurred in the management of microscopic stage III disease due to the data from MSLT-21, the current standard of care for macroscopic stage III nodal malignant melanoma consists of initial surgical treatment with therapeutic lymph node dissection (TLND), followed by consideration for adjuvant therapy consisting of either anti-PD-1 monotherapy2 or BRAF/MEK inhibitors3. This results in improved relapse-free survival, but recurrence is still observed in almost half of the patients within 3-5 years4-6. Preclinical trials7-9 and emerging clinical data10 suggest that neoadjuvant immune checkpoint inhibition may have clinical benefit over adjuvant approaches. Given increasing enthusiasm for adjuvant and neoadjuvant approaches, the OpACIN (NCT02437279, phase I) and OpACIN-neo (NCT02977052, phase II) studies were established to investigate the safety and efficacy of neoadjuvant treatment with immune checkpoint inhibitors (ICI) combination, and to establish optimal dosing regimens to maximize clinical benefit while minimizing toxicity in patients with stage III melanoma. The OpACIN and OpACIN-neo trials

Can Clostridium Difficile infection be prevented?
Jason Xiao1, John Alverdy11Department of Surgery, University of Chicago Medicine, Chicago, IL, 60637
Paper for discussion: Fachi JL, Felipe JS, Pral LP, Silva BK, Correa RO, Cristiny M et al.Butyrate Protects Mice from Clostridium difficile-Induced Colitis through an HIF-1-Dependent Mechanism. Cell Rep. 2019 Apr 16; 27: 750-761.e7. Although rare, Clostridium difficile-induced diarrhoea or colitis can complicate what otherwise appeared to be an uneventful elective operation. This rare, but potentially lethal complication results from multiple factors inherent to performing surgery, such as prolonged periods of starvation, antibiotic exposure, major physiological stress, and sleep deprivation1. C. difficile spores can spread easily, can resist multiple methods of decontamination and can remain viable for long periods of time. In many cases, the bacteria can remain hidden within the host’s gut microbiome and transferred to the healthcare setting by the patient themselves, rather than vice versa. While prevention is the best treatment, C. difficile infections (CDI) often prove resistant to antibiotics, and other modalities may be needed to restore homeostasis to the gut microbiome. Although faecal microbiota transplant has been proposed as a method for both prevention and treatment of CDI, even when severe colitis is present, many believe the most important action of the microbiome is to preserve its ability to produce key multifunctional metabolites 2. For example, the ability of the microbiota to produce the short-chain fatty acids (SCFAs) acetate, propionate, and butyrate has been identified to be an important therapeutic aspect in the prevention and treatment of CDI. SCFAs are absorbed by host intestinal epithelial cells (IECs) and participate in several immunoregulatory roles that influence the host response to inflammation and infection. Past studies have detected reduced SCFA concentrations, particularly butyrate, in patients with CDI3. Elevation of butyrate via dietary modulation or provision of SCFA-producing bacteria has been shown to attenuate CDI severity in animal studies4,5.
Copied!
Connect

Copyright © 2026 River Valley Technologies Limited. All rights reserved.



.png)





.jpg)



