Normal View Dyslexic View
Faecal microbiota transplantation unravelled
Article Review
Schmidt TSB, Li SS, Maistrenko OM, et al. Drivers and determinants of strain dynamics following fecal microbiota transplantation. Nature Medicine (2022). 28(9), 1902-1912
Faecal microbiota transplantation (FMT) is currently recommended in the UK by the National Institute for Health and Care Excellence (NICE) for the treatment of recurrent Clostridioidesdifficile infection (rCDI)1. Additionally, FMT is being extensively investigated for its potential application in surgical diseases, like the prevention of anastomotic leak2, as well as in ulcerative colitis (UC) and its related surgical complications, such as pouchitis3. Modulation of the faecal microbiome has significant implications for surgeons, as emerging evidence suggests pre-operative optimisation of the gut microbiota enhances patient outcomes and reduces surgical risks, such as surgical site infection, postoperative ileus and anastomotic leak4, 5, 6.
However, the mechanisms underlying how FMT has therapeutic benefits remain largely unclear, and accurate outcome predictions are elusive. This remains a fundamental barrier to the wider adoption of this therapy in clinical practice. The recent analysis by Schmidt et al. is therefore of significant importance, as it provides highly novel insights into how FMT works. They conducted a comprehensive meta-analysis investigating the determinants of FMT response in ten different diseases, focusing on strain dynamics and the ecological response, and specifically on donor-recipient microbiota interactions. The researchers assessed strain-level colonisation patterns, resilience, coexistence, and turnover of recipient strains after FMT, developing cross-validated models to predict FMT outcomes by linking significant clinical variables to ecological processes. Their study found that factors of the recipient’s microbiome and donor-recipient microbiome complementarity were primary drivers of both strain dynamics and FMT efficacy.
To investigate the strain dynamics of the donor and recipient microbiome, the authors conducted a meta-analysis on 1,492 faecal metagenomes obtained from 316 FMTs, pre- and post-FMT, used for ten different diseases, including rCDI and UC. They discovered a change in the recipient’s microbiota towards new strains or strains which were at undetectable levels pre-FMT, rather than full rejection or conversion to donor microbiota. This change to new strains was attributed by the authors to the FMT-induced disturbance of the recipient microbiota, rather than the FMT’s content. Donor strain takeover and recipient strain persistence were observed, but only in species that were exclusively present in either the donor’s or recipient’s pre-FMT microbiota. When species were detected in both donor and recipient pre-FMT, coexistence of strains was most common. While donor and new strain takeover were characteristic in certain diseases (mostly UC and rCDI), interestingly, clinical outcomes did not correlate with strain-level changes in any disease. This lack of correlation extended to the metabolome, where increased levels of short-chain-fatty-acid (SCFA) producing bacteria post-FMT also didn’t correlate with clinical outcomes.
To identify factors associated with colonisation outcomes, the authors trained predictive machine learning models using cross-validated Lasso-regularised linear regression. Moderate accuracy was achieved for predicting post-FMT coexistence and recipient strain persistence (LASSO R2=0.58 and 0.49, respectively), while donor colonisation (R2=0.34) and pre-FMT recipient strain resilience (R2=0.35) showed lower variation. Recipient factors or donor-recipient microbiome complementarity were more predictive than donor factors. Recipient species richness and abundance of selected species pre-FMT were important predictors of strain-level outcomes. Interestingly, no individual species’ abundance post-FMT was strongly associated with colonisation outcomes. Successful colonisation of specific species in the recipient, such as B. uniformis, B. vulgatus, and several Oscillospiraceae and Lachnospiraceae species, was highly predictive of donor strain colonisation, suggesting these species may serve as indicators of successful engraftment.
The authors then investigated strain dynamics within each species post-FMT by focusing on 307 species detected in over 50 allogenic FMTs. They observed no consistent patterns of colonisation or persistence and instead found a strong propensity toward donor-recipient strain coexistence, independent of initial strain abundances. Strain-level FMT outcomes varied within each major taxonomic group. Facultatively aerobic species colonised less successfully, while strains with specific metabolic pathways showed higher colonisation success. The authors furthermore found moderate accuracy in predicting resilience of the recipient’s microbiome, donor colonisation, and takeover of donor microbiome. However, high accuracy was observed in predicting recipient strain turnover across almost all species, suggesting that recipient strain displacement may generally be a more deterministic process.
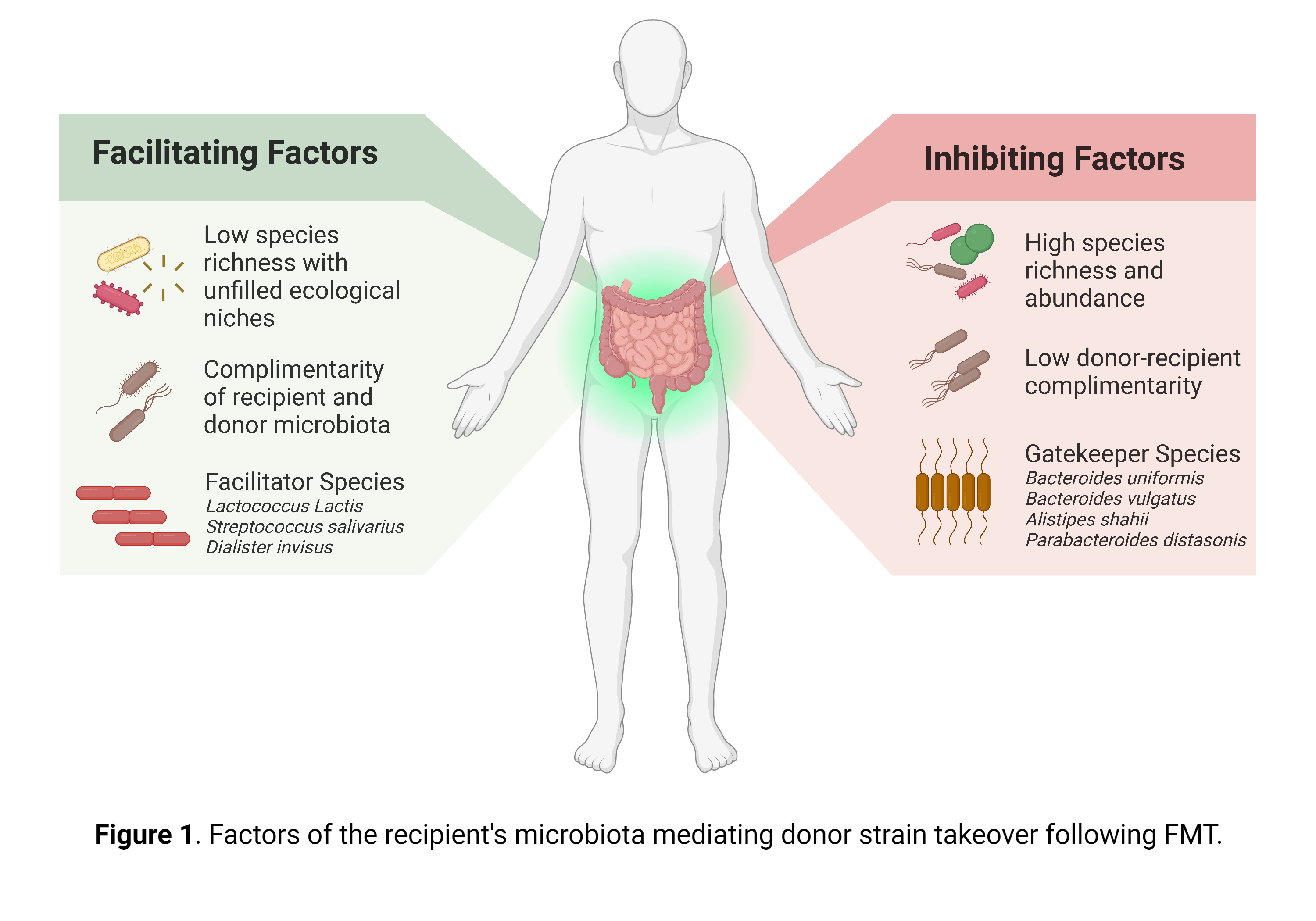
They further demonstrated that species-specific strain dynamics were primarily driven by the recipient microbiome, observing exclusion and facilitation effects. The majority of interactions were inhibitive, with an abundance of specific species in the recipient correlating negatively with the engraftment of colonisation of donor bacteria. Exclusion effects were furthermore stronger for the recipient’s resident community than the donor’s. Colonisation inhibition by the recipient’s microbiome was phylogenetically concentrated, being more common between related species. Bacteroidales species in the recipient microbiota, particularly B. uniformis, B. vulgatus, Alistipes shahii, and Parabacteroides distasonis, were among the strongest colonisation inhibitors and classified by the authors as possible ‘gatekeeper species’.
In contrast, Lactococcus lactis, Streptococcus salivarius, and Dialister invisus in the recipient were the primary colonisation facilitators, typically affecting phylogenetically distant species. Only a few notable predictive species were observed in the donor microbiota, such as B. vulgatus and Evtepia gabavorous. Facilitation and inhibition effects of donor species were generally limited and less predictive of colonisation success, indicating that the donor microbiota has a minimal impact on colonisation outcomes.
However, these species-specific patterns of turnover were superseded by microbiome-level features. Two critical microbiome-level elements accurately predicted strain turnover: pre-FMT species richness in the recipient and donor-recipient dissimilarity. The latter emerged as the most potent FMT outcome predictor, driven by the recipient’s microbiome. A complementary donor microbiome facilitated donor colonisation, but a diverse recipient microbiome remained resilient to colonisation even with high complementarity. Successful colonisation primarily involved donor species occupying an empty niche in the recipient microbiome. Overall, donor microbiome composition exerted a minimal impact on species-specific strain dynamics. These dynamics are especially interesting, as the dissimilarity between diseased and healthy control microbiota has been recognised as a hallmark for numerous FMT indications.
The authors concluded by emphasising that the recipient’s microbiota primarily drives FMT outcome, while the donor microbiota’s role remains ambiguous, see figure 1 for a summary of the recipient’s microbiome in driving donor strain takeover. They further conclude that rCDI and UC were the most responsive conditions to FMT, as treatment led to increased donor bacteria colonisation and clinical response rates, however, as both diseases are often preceded by antibiotic treatment, this should be considered with care. The authors additionally found no evidence supporting that any species is intrinsically more invasive or resilient. Rather, structured species interactions determine outcomes and can be predicted to some extent, with recipient strain turnover being well-predicted, but donor strain takeover not.
This meta-analysis is limited, however, by its focus on the bacterial microbiome and underrepresentation of certain conditions in their dataset. The authors furthermore acknowledge that colonisation may be influenced by variables not examined in this study, notably viral, eukaryotic microbiota, or recipient immunological factors, as well as medication or viability of donor anaerobes following FMT. Finally, the authors propose that future FMT candidates should be stratified based on microbiome richness and ‘gatekeeper’ species, while donor selection should only be taken into consideration for its complementarity with the recipient’s microbiome.
Baseline recipient microbiome diversity and donor-recipient complementarity have been previously theorised to affect FMT outcome, although much of the literature focuses on immunological compatibility7. This meta-analysis offers insight into the role of the microbiota itself on donor-recipient compatibility. Schmidt et al. ecological approach to FMT builds on the work of Gibbons et al. (2020)8, who hypothesized that the evolution of the gut microbiota might be modelled after that of natural ecosystems. The indicator species identified by Schmidt et al. parallel the pioneer species necessary for other species to thrive in larger-scale ecosystems.
However, the lack of relationship between strain dynamics and clinical outcome in all diseases examined in this meta-analysis, including rCDI and UC, suggests the mechanism behind successful treatment with FMT remains obscure. Nevertheless, the importance of the recipient microbiome repeatedly demonstrated in this article builds a strong foundation for the adoption of a more recipient-centred approach over a donor-centred one. Moving away from a search for ‘super-donors’ with an ideal microbiome signature in favour of donors with high donor-recipient complementarity could achieve higher FMT response rates. This means that clinical studies of FMT must move towards detailed phenotyping of the recipient microbiome before any surgical intervention. This is likely to become increasingly important as trials of FMT in surgical diseases become more prevalent.
References
NICE. Faecal microbiota transplant for recurrent Clostridium difficile infection | Guidance and guidelines. https://www.nice.org.uk/guidance/mtg71 (accessed 7 April 2023).
Hajjar R, Gonzalez E, Fragoso G, Oliero M, Alaoui AA, Calvé A, et al. Gut microbiota influence anastomotic healing in colorectal cancer surgery through modulation of mucosal proinflammatory cytokines. Gut. 2022. doi: 10.1136/gutjnl-2022-328389.
Stallmach A, Lange K, Buening J, Sina C, Vital M, Pieper DH. Fecal microbiota transfer in patients with chronic antibiotic-refractory pouchitis. Am J Gastroenterol. 2016 Mar;111(3):441-3. doi: 10.1038/ajg.2015.436.
Duff SE, Battersby CLF, Davies RJ, Hancock L, Pipe J, Buczacki S, et al. The use of oral antibiotics and mechanical bowel preparation in elective colorectal resection for the reduction of surgical site infection. Colorectal Dis. 2020;22(4):364-72. doi: 10.1111/codi.14982.
Paine H, Jones F, Kinross J. Preparing the Bowel (Microbiome) for Surgery: Surgical Bioresilience. Clin Colon Rectal Surg. 2023;36(02):138-45. doi: 10.1055/s-0042-1760675.
Bartolini I, Risaliti M, Ringressi MN, Melli F, Nannini G, Amedei A, et al. Role of gut microbiota-immunity axis in patients undergoing surgery for colorectal cancer: Focus on short and long-term outcomes. World J Gastroenterol. 2020;26(20):2498-513. doi: 10.3748/wjg.v26.i20.2498.
Hanssen NMJ, de Vos WM, Nieuwdorp M. Fecal microbiota transplantation in human metabolic diseases: From a murky past to a bright future? Cell Metab. 2021 Jun 1;33(6):1098-1110. doi: 10.1016/j.cmet.2021.05.005.
Gibbons SM. Keystone taxa indispensable for microbiome recovery. Nat Microbiol. 2020 Sep;5(9):1067-1068. doi: 10.1038/s41564-020-0783-0. Erratum in: Nat Microbiol. 2020 Oct;5(10):1307.




.png)





.jpg)




